
Concept explainers
(a)
Interpretation:
The
Concept introduction:
Nuclear magnetic resonance is a type of spectroscopy in which number of different kind of protons present in different environment can be detected. The
Answer to Problem 13.13P
The
Explanation of Solution
The structure of
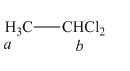
Figure 1
In the given structure of
The
(b)
Interpretation:
The
Concept introduction:
Nuclear magnetic resonance is a type of spectroscopy in which number of different kind of protons present in different environment can be detected. The
Answer to Problem 13.13P
The
Explanation of Solution
The structure of
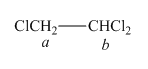
Figure 2
In the given structure of
The
(c)
Interpretation:
The
Concept introduction:
Nuclear magnetic resonance is a type of spectroscopy in which number of different kind of protons present in different environment can be detected. The
Answer to Problem 13.13P
The
Explanation of Solution
The structure of
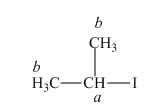
Figure 3
In the given structure of
The
(d)
Interpretation:
The
Concept introduction:
Nuclear magnetic resonance is a type of spectroscopy in which number of different kind of protons present in different environment can be detected. The
Answer to Problem 13.13P
The
Explanation of Solution
The structure of
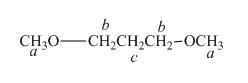
Figure 4
In the given structure of
The
(e)
Interpretation:
The
Concept introduction:
Nuclear magnetic resonance is a type of spectroscopy in which number of different kind of protons present in different environment can be detected. The
Answer to Problem 13.13P
The
Explanation of Solution
The structure of oxetane ring is shown below in Figure 5.
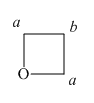
Figure 5
In the given structure of oxetane ring, two types of hydrogens are present. The hydrogen
The
(f)
Interpretation:
The
Concept introduction:
Nuclear magnetic resonance is a type of spectroscopy in which number of different kind of protons present in different environment can be detected. The
Answer to Problem 13.13P
The
Explanation of Solution
The structure of
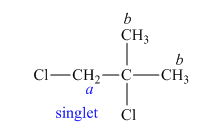
Figure 6
In the given structure of
The
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry
- 9) Deduce the structure of an unknown compound with molecular formula C5H80 using information given by its infrared spectrum Frequency (cm-1): 3100, 2950, 2800, 1695, 1610,1450, 1375 HOarrow_forwardwhat is the bond line formula of C6H15N with respect to given IR spectra and NMRarrow_forward8. Which of the following pairs of compounds is likely to absorb radiation at the longer wavelength and with greater intensity? (a) CH3CH2CO₂H or CH2=CHCO₂H (b) CH3CH=CHCH=CHCH3 or CH3C = C—C = CCH3 OCH3 (c) or CH3arrow_forward
- Predict how the bond, shown in each of the following compounds will break with the aidof the appropriate arrow(s). Also give the structure(s) of the species formed in eachreaction.(a) CH3── I →(b) Q ── Q →arrow_forwardBelow are two molecules, cyclohexylamine (A) and aniline (B). When analyzed by Infrared Spectroscopy (IR) two different frequencies are observed for the C-N absorption band. Which molecule will have the lower wavenumber absorption band for the C-N bond. Explain why in 35 words or less. „NH2 NH2 А Barrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energyarrow_forward
- The pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energy. (continued)762 CHAPTER 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy(e) Show how many electrons are in each MO for the pentadienyl radical (ground state).(f) Show how your molecular orbital picture agrees with the resonance picture showing delocalization of the unpairedelectron onto three carbon atoms.(g) Remove the highest-energy electron from the pentadienyl radical to give the pentadienyl cation. Which carbon atomsshare the positive charge? Does this picture agree with the resonance picture?(h) Add an…arrow_forwardThe analysis of the structure of compound X which has the molecular formula C14H10O2 provides spectroscopic data as shown below. By systematic analysis, determine the structure of the compound!arrow_forwardThe NMR spectrum of bromocyclohexane indicates a low field signal (1H) at δ 4.16. To room temperature, this signal is a singlet, but at -75 ° C it separates into two peaks of unequal area (but totaling one proton): δ 3.97 and δ 4.64, in ratio 4.6: 1.0. How do you explain the doubling in two peaks? According to the generalization of the previous problem, what conformation of the molecule predominates (at -75 ° C)? What percentage of the molecules does it correspond to? Solve all parts otherwise down vote and hand written solutionarrow_forward
- A compound of formula C5H11Br has the following 1H NMR characteristics: Singlet,6H; Triplet,3H; Quartet,2H. Provide a structure for the compound consistent with this information. Support your answer.arrow_forwardBased on this IR spectrum and 1H NMR spectrum and the given chemical formula is C9H10O2, what would be the chemical structure of the compound? Please give a full analysis of the NMR spectra for given compound.arrow_forwardDetermine the weakest C-H bond in each of the following compounds. (a) (b) (c) ОНarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





