
(a)
The moment of inertia and rotation energy.
(a)
Answer to Problem 31P
The moment of inertia is
Explanation of Solution
Given:
The equilibrium separation is
Formula used:
The expression for moment of inertia is given by,
The expression for rotational energy is given by,
Calculation:
The moment of inertiais calculated as,
The rotational energy is calculated as,
Conclusion:
Therefore, the moment of inertia is
(b)
The energy level diagram.
(b)
Answer to Problem 31P
The energy level diagram is shown in figure 1.
Explanation of Solution
Calculation:
The energy level diagram for the rotational level from
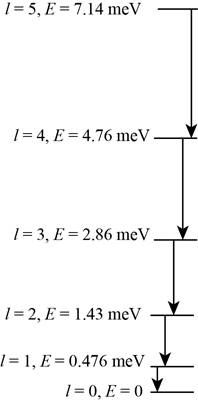
Figure 1
Conclusion:
Therefore, the energy level diagram is shown in figure 1.
(c)
The wavelength for each transition of part (b).
(c)
Answer to Problem 31P
The wavelengths for each transition from start are
Explanation of Solution
Formula used:
The expression for wavelength is given by,
Calculation:
The first wavelength is calculated as,
The second wavelength is calculated as,
The third wavelength is calculated as,
The fourth wavelength is calculated as,
The fifth wavelength is calculated as,
Conclusion:
Therefore, the wavelengths for each transition from start are
Want to see more full solutions like this?
Chapter 37 Solutions
Physics for Scientists and Engineers
- To determine the equilibrium separation of the atoms in the HCl molecule, you measure the rotational spectrum of HCl. You find that the spectrum contains these wavelengths (among others): 60.4 mm, 69.0 mm, 80.4 mm, 96.4 mm, and 120.4 mm. (a) Use your measured wavelengths to find the moment of inertia of the HCl molecule about an axis through the center of mass and perpendicular to the line joining the two nuclei. (b) The value of l changes by +-1 in rotational transitions. What value of l for the upper level of the transition gives rise to each of these wavelengths? (c) Use your result of part (a) to calculate the equilibrium separation of the atoms in the HCl molecule. The mass of a chlorine atom is 5.81 * 10-26 kg, and the mass of a hydrogen atom is 1.67 * 10-27 kg. (d) What is the longest-wavelength line in the rotational spectrum of HCl?arrow_forwardThe frequency of the photon that causes the υ = 0 to υ = 1 transition in the CO molecule is 6.42 x 1013 Hz. We ignore any changes in the rotational energy for this example.(A) Calculate the force constant k for this molecule. (B) What is the classical amplitude A of vibration for this molecule in the υ = 0 vibrational state?arrow_forwardWhat is the energy of a photon emitted from a CO molecule that transitions from the rotational Ji to Ji-1 level. Express your answer in units of ℏ^2/I, so you just have an integer value as a final answer. Values: Ji = 3 The equation I have in the teacher's ppt is E_rotational = (J*(J+1)*ℏ^2)/(2*I)arrow_forward
- Consider a CO molecule that is initially in the ground state of n = 0, l = 0. If the energy of a vibrational transition from the n = 0 state to the n = 1 state in CO could instead be absorbed in a rotational transition, what would be the value of l for the final state?arrow_forwardThe effective spring constant associated with bonding in the N2 molecule is 2 297 N/m. The nitrogen atoms each have a mass of 2.32 x 10-26 kg, and their nuclei are 0.120 nm apart. Assume the molecule is rigid. The first excited vibrational state of the molecule is above the vibrational ground state by an energy difference ΔE. Calculate the J value of the rotational state that is above the rotational ground state by the same energy difference ΔE.arrow_forwardIn a vibrational-rotational spectroscopy the total energy is the sum of the energies coming from the vibration and rotation (E = E + E₁). Selection rule suggests that for transition to occur Av = ±1 and Al = ±1. At room temperature, it is assumed that the lowest vibrational state is populated and the energy can only travel upwards due to lack of population of upper vibrational states thus Av = +1. What would be the energy of a line for R, P and Q-branch if a.) Al = +1, b.) Al = -1 and c.) Al = 0 respectively.arrow_forward
- This problem deals with the splitting of rotational energy levels of diatomic molecules. If one atom of the molecule has more than one stable isotope, then both isotopes are normally present in a sample. Show that the fractional change ∆f/f in the observed frequency of a photon emitted in a transition between adjacent rotational states is equal to the fractional difference in the reduced mass ∆μ/μ for molecules containing the two different isotopes.arrow_forwardIn solid KCI the smallest distance between the centers of a. potassium ion and a chloride ion is 314 pm. Calculate the length of the edge of the unit cell and the density of KCI, assuming it has the same structure as sodium chloride.arrow_forwardThe moment of inertia of water molecule about an axis bisecting the HOH angle is1.91x10-47 kg m2. Its minimum angular momentum about that axis (other than zero) is ℏ. Inclassical terms, how many revolutions per second do the hydrogen atoms make about the axiswhen in that state? Calculate the rotational constant (cm-1) and bond length of H2O. Does the bondlength seem reasonable?arrow_forward
- We can apply the rigid rotor approximation to the rotational energy levels of a diatomic molecule, with E, = BJ(J + 1), where B represents a rotational constant. (a) Given the selection rules for ro-vibrational transitions, develop a formula for an absorption in the R branch from a state with angular momentum J. Assume a harmonic oscillator vibration with frequency 785 cm¹ originating in the ground vibrational state (i.e. n = 0). You will be given a value for B and asked to compute the energy for a particular transition on the submission quiz. (b) Suppose you measure several R-branch rotational transitions for 24Mg¹60, and you find that the absorption lines are uniformly spaced 1.1485 cm-¹ apart. The quantity, B, represents a rotational constant related to the moment of inertia, I, of the molecule, via h B = 8π² cl when expressed in wavenumber (cm-¹) units. Based on this information, what is the value of the moment of inertia (in kg-m²) for MgO? (c) Using the results you have found,…arrow_forwardN 2 has a molecular weight of 28.02 g/mol a bit larger than that of a Ne atom, 20.18 g/mol. (a) At a particular temperature, Z trans= 1.90 x 10 26 for Ne in a specific container. What is the translational partition function for a N2 molecule in this container at the same temperature? (b) At 100 K, the rotational partition function for N2is found to be 17.39. What would you expect it to be at 500 K?arrow_forwardWhen a hypothetical diatomic molecule having atoms 0.8860 nm apart undergoes a rotational transition from the l = 2 state to the next lower state, it gives up a photon having energy 8.841 * 10-4 eV. When the molecule undergoes a vibrational transition from one energy state to the next lower energy state, it gives up 0.2560 eV. Find the force constant of this molecule.arrow_forward
 Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning

